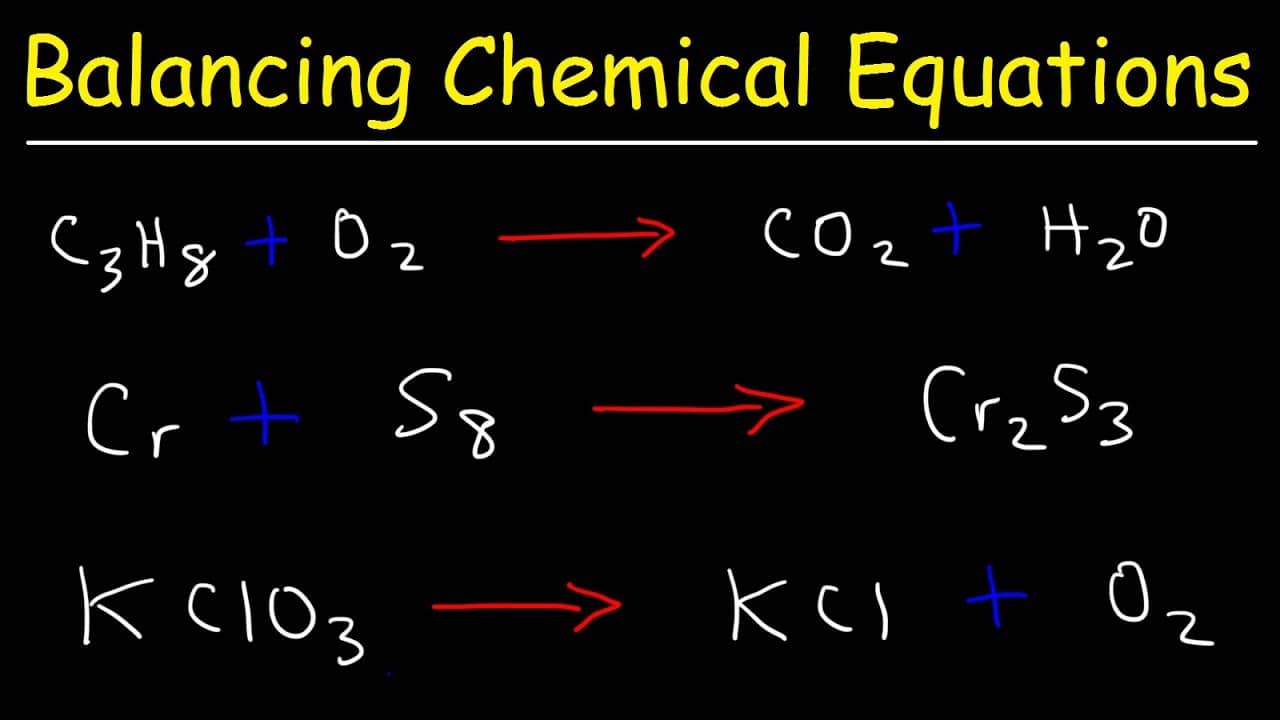
Here we have five equations for four unknowns, however, the last one is dependent on the fourth, so it can be omitted. They will form a system of linear equations: Then we write the balance equations for each element in terms of the unknowns: We start by introducing unknown coefficients: Let me illustrate this method by example. Therefore this method could be used for any type of chemical reaction (including redox reactions). So, you just need to create a set of algebraic equations expressing the number of atoms of each element involved in the reaction and solve it. Balancing chemical equations is the process of ensuring the conservation of matter. Therefore, the number of each type of atom on each side of a chemical equation must be the same. The algebraic method is based on the Law of Conservation of Mass – that matter can neither be created nor destroyed. This chemical equation balancer uses the algebraic method – which is usually quite complex for manual calculations, however, it fits the computer program perfectly. The last two are used for redox reactions. Ion-electron method, or half-reaction method.Inspection method, or "hit & trial" method.I would love to know which of the three methods you prefer.There are several methods of balancing chemical equations: So, now that you’ve gone through the basics, how about testing out your skills? Each level below has 5 questions. I’ll walk you through the 4 steps involved in this video: Basically, what you’ll do is set up a bunch of equations and then solve them to get the coefficients (numbers in front of the formula). The final method is for those who loves solving algebraic equations or when you’re trying to balance complex equations (like the ones in Ninja level – see practice questions below). There are 4 simple steps in balancing equation using this method. It pretty much works like the first method, however, you keep track of the changes by writing them down. We’ll try out the second method on this new equation. You can take my word for it, or give it a try and see how long it’ll take you to balance this equation by inspection. However, it gets taxing when you encounter more complicated equation like this: C 6H 5F + O 2 → CO + H 2O + F 2. This is a rather popular method and it works pretty well to a certain extent. Since we added a 2 in front of NaCl, our Na on the right-hand side is no longer balanced, so we’ll need to add a 2 in front of NaF as well. So, we’ll place a 2 in front of NaCl like this: Looks like it’s not balanced since there’s one on the left but two on the right. Na is balanced since there is one on the left-hand side and one on the right-hand side. You balance the equation by inspecting it. Personally, I prefer Method 2 when it comes to solving easy to intermediate equations because it is simple and doesn’t tax my brain too much. There are 3 methods in mastering this art. If you count the number of atoms on both sides, you’ll notice that they are not equal: The compounds on the left-hand side (before the arrow) are called reactants while the compounds on the right-hand side (after the arrow) are called products. So the main goal of balancing chemical equation is to get the number of atoms to be the same on each side of the equation. You’ll be able to get the molar ratios for all the compounds involved in the reaction and that will enable you to perform various types of calculations correctly (more on that in future posts). Jokes aside, being able to balance chemical equation will open up a lot of possibilities. Is it important to be able to balance chemical equations correctly and quickly? You bet! Why so? First of all, you’ll make your chemistry teacher happy and maybe impress someone else along the way.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
